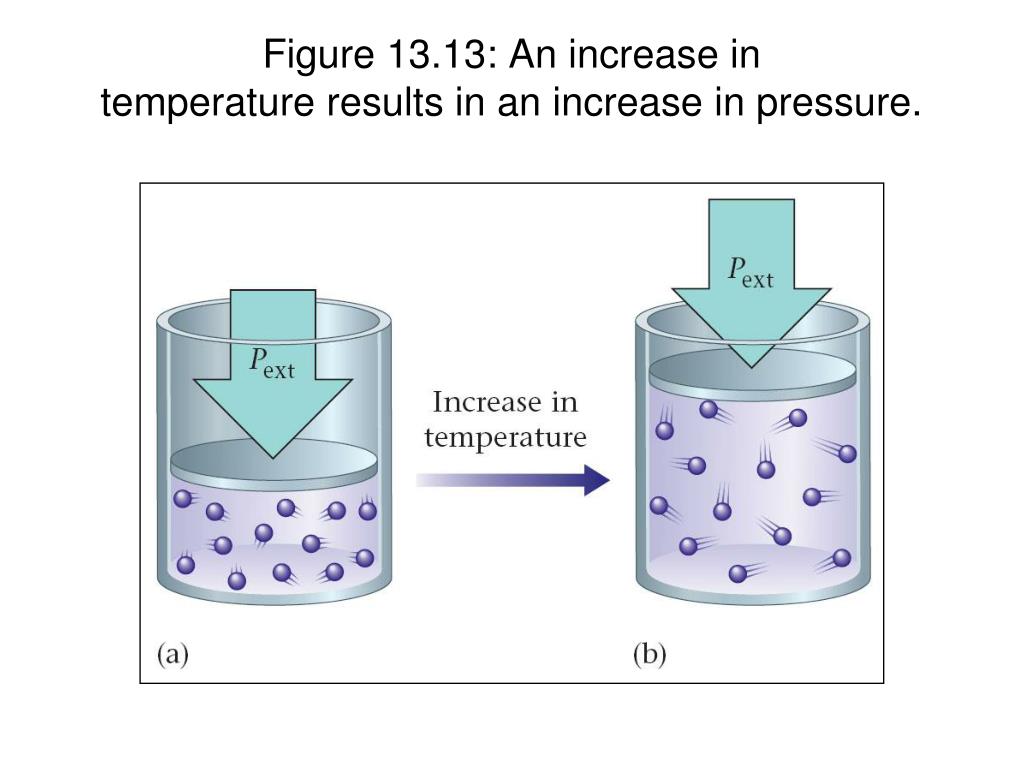
Does Partial Pressure Increase With Temperature . — the vapor pressure of a substance depends on both the substance and its temperature—an increase in. if you're seeing this message, it means we're having trouble loading external resources on our website. If you're behind a web filter, please make. — the partial pressure of a gas is the pressure that gas would exert if it occupied the container by itself. The pressure exerted by each. — the law used to find partial pressure assumes the temperature of the system is constant and the gas behaves as an ideal gas, following the ideal. — the partial pressure of each gas in a mixture is proportional to its mole fraction.
from www.slideserve.com
— the partial pressure of a gas is the pressure that gas would exert if it occupied the container by itself. The pressure exerted by each. — the vapor pressure of a substance depends on both the substance and its temperature—an increase in. — the law used to find partial pressure assumes the temperature of the system is constant and the gas behaves as an ideal gas, following the ideal. If you're behind a web filter, please make. if you're seeing this message, it means we're having trouble loading external resources on our website. — the partial pressure of each gas in a mixture is proportional to its mole fraction.
PPT Unit 4 Phases of Matter (Chapters 1314) PowerPoint Presentation
Does Partial Pressure Increase With Temperature — the partial pressure of a gas is the pressure that gas would exert if it occupied the container by itself. The pressure exerted by each. — the partial pressure of each gas in a mixture is proportional to its mole fraction. — the partial pressure of a gas is the pressure that gas would exert if it occupied the container by itself. If you're behind a web filter, please make. — the law used to find partial pressure assumes the temperature of the system is constant and the gas behaves as an ideal gas, following the ideal. if you're seeing this message, it means we're having trouble loading external resources on our website. — the vapor pressure of a substance depends on both the substance and its temperature—an increase in.
From www.wikihow.com
How to Calculate Partial Pressure 14 Steps (with Pictures) Does Partial Pressure Increase With Temperature — the partial pressure of a gas is the pressure that gas would exert if it occupied the container by itself. The pressure exerted by each. — the partial pressure of each gas in a mixture is proportional to its mole fraction. If you're behind a web filter, please make. if you're seeing this message, it means. Does Partial Pressure Increase With Temperature.
From www.researchgate.net
Temperature dependence of the equilibrium oxygen partial pressure for Does Partial Pressure Increase With Temperature if you're seeing this message, it means we're having trouble loading external resources on our website. If you're behind a web filter, please make. — the law used to find partial pressure assumes the temperature of the system is constant and the gas behaves as an ideal gas, following the ideal. The pressure exerted by each. —. Does Partial Pressure Increase With Temperature.
From opentextbc.ca
9.2 Relating Pressure, Volume, Amount, and Temperature The Ideal Gas Does Partial Pressure Increase With Temperature — the vapor pressure of a substance depends on both the substance and its temperature—an increase in. If you're behind a web filter, please make. — the partial pressure of a gas is the pressure that gas would exert if it occupied the container by itself. if you're seeing this message, it means we're having trouble loading. Does Partial Pressure Increase With Temperature.
From exoudqbxz.blob.core.windows.net
Does Temperature Increase When Pressure Increases at Hollis Winter blog Does Partial Pressure Increase With Temperature — the partial pressure of each gas in a mixture is proportional to its mole fraction. If you're behind a web filter, please make. The pressure exerted by each. if you're seeing this message, it means we're having trouble loading external resources on our website. — the law used to find partial pressure assumes the temperature of. Does Partial Pressure Increase With Temperature.
From www.researchgate.net
Partial Pressure of water vapor vs. temperature, including saturation Does Partial Pressure Increase With Temperature If you're behind a web filter, please make. The pressure exerted by each. — the partial pressure of each gas in a mixture is proportional to its mole fraction. — the law used to find partial pressure assumes the temperature of the system is constant and the gas behaves as an ideal gas, following the ideal. if. Does Partial Pressure Increase With Temperature.
From dxoithjoo.blob.core.windows.net
Does Pressure Increase With Temperature at Mildred Blose blog Does Partial Pressure Increase With Temperature if you're seeing this message, it means we're having trouble loading external resources on our website. — the vapor pressure of a substance depends on both the substance and its temperature—an increase in. — the partial pressure of each gas in a mixture is proportional to its mole fraction. The pressure exerted by each. If you're behind. Does Partial Pressure Increase With Temperature.
From www.quora.com
If temperature increases, pressure increases. Does temperature increase Does Partial Pressure Increase With Temperature The pressure exerted by each. If you're behind a web filter, please make. — the partial pressure of each gas in a mixture is proportional to its mole fraction. — the partial pressure of a gas is the pressure that gas would exert if it occupied the container by itself. — the vapor pressure of a substance. Does Partial Pressure Increase With Temperature.
From usq.pressbooks.pub
8.5 Transport of Gases Fundamentals of Anatomy and Physiology Does Partial Pressure Increase With Temperature — the law used to find partial pressure assumes the temperature of the system is constant and the gas behaves as an ideal gas, following the ideal. The pressure exerted by each. If you're behind a web filter, please make. — the partial pressure of a gas is the pressure that gas would exert if it occupied the. Does Partial Pressure Increase With Temperature.
From cooljargon.com
Anatomy and Physiology Transport of Gases Does Partial Pressure Increase With Temperature — the partial pressure of each gas in a mixture is proportional to its mole fraction. — the vapor pressure of a substance depends on both the substance and its temperature—an increase in. — the law used to find partial pressure assumes the temperature of the system is constant and the gas behaves as an ideal gas,. Does Partial Pressure Increase With Temperature.
From www.slideserve.com
PPT Gases & colligative properties PowerPoint Presentation, free Does Partial Pressure Increase With Temperature — the law used to find partial pressure assumes the temperature of the system is constant and the gas behaves as an ideal gas, following the ideal. The pressure exerted by each. — the vapor pressure of a substance depends on both the substance and its temperature—an increase in. if you're seeing this message, it means we're. Does Partial Pressure Increase With Temperature.
From chem.libretexts.org
6.3 Relationships among Pressure, Temperature, Volume, and Amount Does Partial Pressure Increase With Temperature — the vapor pressure of a substance depends on both the substance and its temperature—an increase in. — the partial pressure of each gas in a mixture is proportional to its mole fraction. — the partial pressure of a gas is the pressure that gas would exert if it occupied the container by itself. The pressure exerted. Does Partial Pressure Increase With Temperature.
From www.youtube.com
Law of Partial Pressures YouTube Does Partial Pressure Increase With Temperature The pressure exerted by each. if you're seeing this message, it means we're having trouble loading external resources on our website. If you're behind a web filter, please make. — the vapor pressure of a substance depends on both the substance and its temperature—an increase in. — the partial pressure of each gas in a mixture is. Does Partial Pressure Increase With Temperature.
From www.youtube.com
Worked example Calculating partial pressures AP Chemistry Khan Does Partial Pressure Increase With Temperature — the vapor pressure of a substance depends on both the substance and its temperature—an increase in. — the law used to find partial pressure assumes the temperature of the system is constant and the gas behaves as an ideal gas, following the ideal. — the partial pressure of a gas is the pressure that gas would. Does Partial Pressure Increase With Temperature.
From www.slideserve.com
PPT Chapter 14 PowerPoint Presentation, free download ID3552130 Does Partial Pressure Increase With Temperature if you're seeing this message, it means we're having trouble loading external resources on our website. — the law used to find partial pressure assumes the temperature of the system is constant and the gas behaves as an ideal gas, following the ideal. — the partial pressure of a gas is the pressure that gas would exert. Does Partial Pressure Increase With Temperature.
From www.slideserve.com
PPT Unit 4 Phases of Matter (Chapters 1314) PowerPoint Presentation Does Partial Pressure Increase With Temperature — the law used to find partial pressure assumes the temperature of the system is constant and the gas behaves as an ideal gas, following the ideal. if you're seeing this message, it means we're having trouble loading external resources on our website. — the vapor pressure of a substance depends on both the substance and its. Does Partial Pressure Increase With Temperature.
From www.slideserve.com
PPT Dalton’s Law of Partial Pressure PowerPoint Presentation, free Does Partial Pressure Increase With Temperature — the vapor pressure of a substance depends on both the substance and its temperature—an increase in. if you're seeing this message, it means we're having trouble loading external resources on our website. If you're behind a web filter, please make. — the law used to find partial pressure assumes the temperature of the system is constant. Does Partial Pressure Increase With Temperature.
From www.slideserve.com
PPT Gas MixturesPartial Pressure PowerPoint Presentation, free Does Partial Pressure Increase With Temperature if you're seeing this message, it means we're having trouble loading external resources on our website. — the vapor pressure of a substance depends on both the substance and its temperature—an increase in. — the partial pressure of a gas is the pressure that gas would exert if it occupied the container by itself. The pressure exerted. Does Partial Pressure Increase With Temperature.
From studyrocket.co.uk
Turning Forces and Pressure GCSE Physics AQA Revision Study Rocket Does Partial Pressure Increase With Temperature — the partial pressure of a gas is the pressure that gas would exert if it occupied the container by itself. — the partial pressure of each gas in a mixture is proportional to its mole fraction. if you're seeing this message, it means we're having trouble loading external resources on our website. — the vapor. Does Partial Pressure Increase With Temperature.